Together with the Protein Expression Facility (PEF) we recently took delivery of a Refeyn mass-photometry system that is currently located in the newly refurbished QBP CryoEM facility.
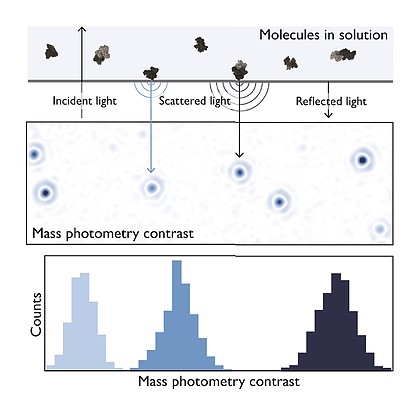
The Refeyn OneMP system applies the principle of interference reflection microscopy and interferometric scattering microscopy to quantify light scattered by a single molecule on a glass surface. The amount of light scattered by each molecule is directly correlated to its molecular mass (literature).
Based on the above principle the Refeyn OneMP can monitor protein-protein interactions at a single-molecule level with high-sensitivity and can at the same time determine molecular weight of proteins and protein complexes with a high dynamic range and great accuracy.
The mass photometer is an ideal tool for quality control in the protein structural analysis workflow as it can assess the molecular mass and the oligomerisation status of a sample in one measurement.
The amount of protein needed for an analysis is very small with only 3µl of protein solution needed at a nano-molar concentration range. After the initial set up, measurement takes in general one minute or slightly longer if required. The MS-Windows based software is very intuitive and only minimal training is needed to become familiar with the Refeyn OneMP.
The CMM QBP CryoEM facility plans to fully integrate mass-photometry as an initial control of the sample quality before it can be observed under cryogenic conditions in the cryoTEMs. PEF has integrated mass-photometry as part of their protein analytics to routinely characterise all purified proteins. Furthermore, mass-photometry is also an independent research tool for protein-protein interaction studies, formation of complexes and formation of protein assemblies etc. as well as interaction dynamics of proteins under various buffer conditions.



